Stem cell therapies have the potential to revolutionize the practice of medicine. Treated with respect, they can help our bodies call forth their own healing mechanisms. Manipulated too far, and they become another potentially carcinogenic drug in the pharmacy. How stem cell therapy is perceived, and what happens to those who bring the therapy from the lab bench to the treatment table, may well determine the course of medical therapy in the foreseeable future.
Low tech or high tech? Use stems cells pretty much as they are found in the body? Or manipulate them chemically, and patent them, and turn them into a drug therapy? Therein lies a great conundrum. And yet... there need not be a war.
Sometimes herbal medicine works very well. Sometimes the body is so ill that herbs are not enough, and pharmaceutical drugs work better in the immediate emergency.
Perhaps it will be the same with stem cells. Sometimes the gentle approach - encouraging stem cells to circulate in the body - will be sufficient to restore health. Sometimes we may need to force them into dividing and multiplying, and even potentially using someone else's stem cells, if the organism is so weak that it cannot provide its own healthy ones.
The ultimate goal is restoration of function and healing of the body.
Tiera 450 – credit: Soler’s Fractal Gallery
Stem cells are the origin of our physical bodies, the cells which have the potential to develop into any organ of the body at any time, given the right circumstances or environment. They are a holograph of our bodies - each stem cell carries within it the image or the potential of the whole. They are formed when the first sperm joins the first egg, and the dance of physical life begins.
What makes them change? Develop into muscles and blood and nerves and a heart?
 This dance of life is finely orchestrated, involving chemical messengers
in just the right
quantity at just the right
time.
This dance of life is finely orchestrated, involving chemical messengers
in just the right
quantity at just the right
time.
Research into stem cells has exploded in the past 50 years. An interesting article published in Research Trends in 2013 states that the number of articles increased by 7% per year after 2006.[1]
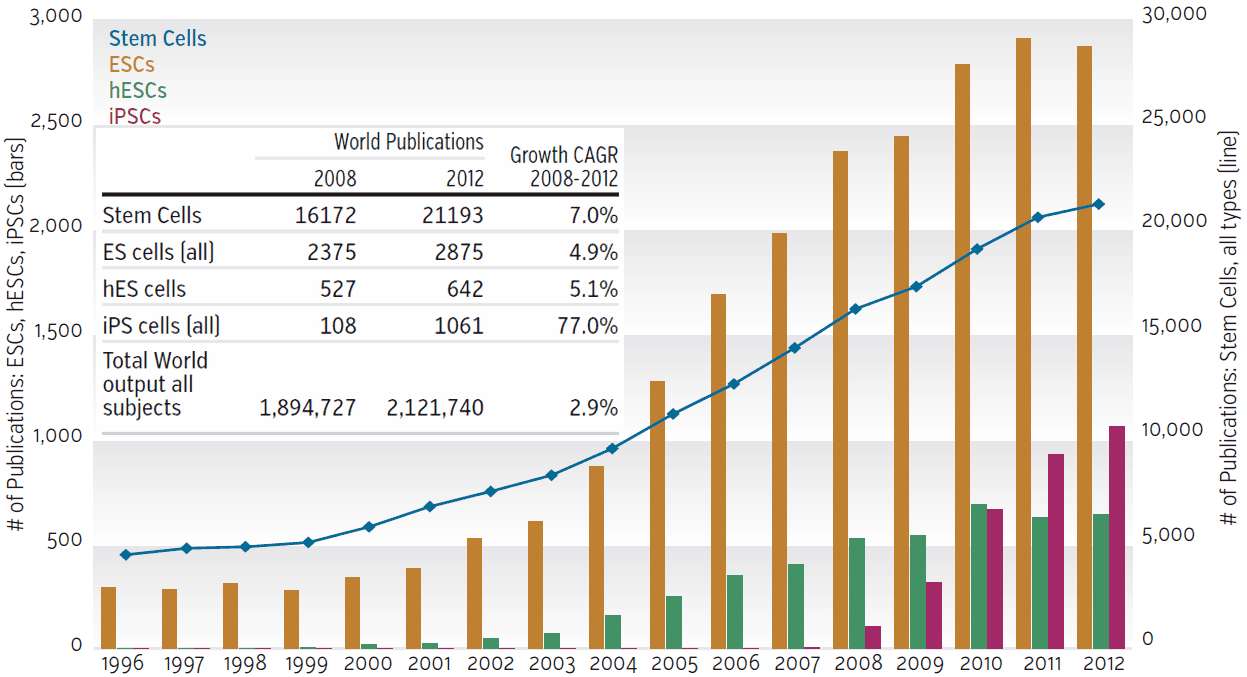
Downloaded 5/30/2015 from http://www.researchtrends.com/issue-36-march-2014/stem-cell-research/
attributed to Scopus.
Wrights/Giemsa stained human embryonic stem cell (hESC) colony on murine embryonic fibroblast feeder cells. The colony contains roughly 50-70 individual hESCs. Photo courtesy of M. William Lensch, PhD. Permission to use requested from willy_lensch@harvard.edu
History
The term "stem cell" first appeared in the scientific literature in 1968, when this term was given by Ernst Haeckel to the prototype unicellular organisms from which all multicellular organisms evolved.[2]
A PubMed search for "stem cells" in 2015 brings up over 100,000 articles. Stem cell therapy clinics have sprung up all over the country - indeed, all over the world - as physicians and patients try to take advantage of their potentially youthful and healing properties.
Stem cells have the ability to hone in on areas of injury, allowing our bodies to heal from wounds.[3]
Stem cells are also the original source of all the tissues in our bodies - which tissues develop depends upon Nature's exquisite sense of timing and the soup of messenger molecules to which the stem cells are exposed. When things go awry, the results may be quite obvious (as in children born with cleft palate, or without a brain). Results may also be less obvious, as in children born with sickle cell trait, or who develop infantile cancers after birth.
Like everything in our experience, there appears to be an optimal level of stem cells. Too many, and we have what we diagnose as childhood leukemia. Too few, and we have grossly abnormal development and childhood death.
In 1981 the existence of embryonic stem cells was discovered - in mice.[4]
In 1968 the first successful bone marrow transplant procedure was performed.[5]
Initial work with stem cells involved bone marrow transplantation for treatment of leukemia - in the thought that if the entire bone marrow supply was destroyed by radiation, it should be possible to restore it using healthy bone marrow.[6] Since the patient's own bone marrow was the source of the illness, transplanted bone marrow was by necessity taken from someone else (heterologous). Risks were many, and patients not infrequently died of overwhelming infection before the new bone marrow could become established.
We gradually came to realize that bone marrow is not the only source of stem cells.
More recently there has been a furor of activity in the area of stem cells - both research and clinical use. Stem cells have been isolated from peripheral blood, from many different organs, and from the adipose tissue - that body fat which we have in such abundance, and that we are always anxious to get rid of.
For an interesting lecture on basic stem cell theory, here is a lecture by Hazel Sive at MIT. https://www.youtube.com/watch?v=62FdhX-zS2Y.[7]
For a more advanced lecture on the uses of pluripotent stem cells, here is a lecture by Robert Lanza, MD on photoreceptors in the eye, and their clinical trial using stem cells to treat macular degeneration. https://www.youtube.com/watch?v=4B8U7AfnbXk
Other sources of stem cells have been discovered and utilized over the ensuing years.
It was discovered that embryonic stem cells were pleuripotent - not a big surprise, since obviously the embryo has to have some way of manufacturing all the various tissues and organs that it needs in order or it would never grow up to become a living being. There was of course a battle, with the lines being drawn between the scientists and those who accused them of being baby-killers. Eventually in 2001 President George W Bush signed an order permitting the use of embryonic stem cell lines but not of new embryonic stem cells. To this day the battle continues, with embryonic stem cells still being exported from some countries and used in stem cell treatment facilities around the world. These embryonic cells do not have the same protective effect on injured lungs that bone marrow derived cells have.[8] There is sufficient controversy over when life begins that, in the absence of compelling reasons to use these cells, it seems unnecessary to use this source.
Stem cells have been found in so many different organs and tissues since that time that in 2006 the International Society for Cellular Therapy set minimum criteria to define human mesenchymal stem cells.[9] First, when cultured in a petri dish, they are adherent to the sides of the dish; second, they express specific cell markers (CD105, CD73 and CD90) and lack specific other markers (long list, including HLA surface markers) and third, they are capable of so-called cell lineage differentiation - they can turn into bone, fat and cartilage cells when cultured.
Stems cell have been found not only in bone marrow, but also in all the following tissues: amniotic fluid, placenta, umbilical cord, muscle, adipose tissue, peripheral blood, skeletal muscle, connective tissue.
Bone marrow contains a population of stem cells which can differentiate into bone, cartilage, tendon, muscle, fat and connective tissue. Bone marrow derived stem cells (BMSC) decrease in number and differentiation capacity with age. Bone marrow is pretty easy to obtain - the procedure is performed in hospital oncology clinics every day in almost every hospital in the country.
Umbilical cord blood is a rich source of stem cells which can differentiate into bone, cartilage, fat, heart and nerve tissues. Collection of cord blood is easy to accomplish. If transplanted into a different person, these cells are less likely to cause a bad immune reaction (called "graft vs host") because the cells are immature. Of course when they are taken from the umbilical cord and placenta, the baby to whom they originally belonged does not get the benefit.
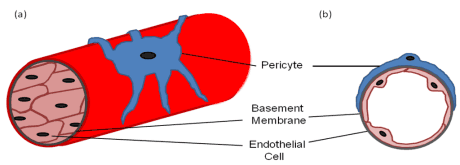
(a) A diagram of a longitudinal section of a blood vessel covered by a pericyte; (b) A cross-sectional diagram of a blood vessel showing the endothelial cell and pericyte interaction with a common basement membrane. Used by permission from the National Library of Medicine – Pericytes, Mesenchymal Stem Cells and the Wound Healing Process. Mills SJ, Cowin AJ, Kaur P – (2013)
Adipose-derived mesenchymal stem cells are, as the name suggests, derived from the fatty tissues of our body.
They are easily collected using a well-established procedure, liposuction,
and can be isolated and given back to the patient after minimal processing.
They do not express Major
 Histocompatibility Class II (MHC) surface markers and as such are invisible
to the immune system.
Histocompatibility Class II (MHC) surface markers and as such are invisible
to the immune system.
All mesenchymal stem cells are pericytes. Pericytes for years were thought to be simply responsible for maintaining blood vessel integrity. We now know that they serve a far larger function. They have stem-cell properties, and are able to differentiate into multiple cell types. They are essential for wound healing.[10]
In recent years, stem cells have been used therapeutically for a multitude of conditions, by a multitude of different routes, extracted, often cultured to multiply them before re-injecting them.
One excellent review article maintains: "Current evidence suggests that tendinopathies arise from a multivariate etiology. It is increasingly evident that intrinsic mechanisms play a greater role than extrinsic mechanisms in this process."[11]
Joint, tendon and rotator cuff injections are probably the easiest and most immediately visible success story in the world of stem cell therapy. Articles have appeared in the lay press since 2011 detailing the use of stem cell therapies by athletes to repair injuries - reportedly with good success.[12]
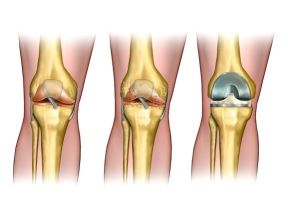
Healthy knee anatomy, degenerative arthritis of the knee and replacement surgery. Digital illustration.
Joints are mostly easily accessible with a needle. Joint injections do
not require major surgery or general anesthesia. It is easy to get medicine
(steroids, for instance) and/or stem cells and/or
 platelet rich plasma and/or a fat cushion into them, and the risk of complications
is low, as long as aseptic technique is used for the injection procedure.
platelet rich plasma and/or a fat cushion into them, and the risk of complications
is low, as long as aseptic technique is used for the injection procedure.
Chronically inflamed joints appear to have fewer stem cells at the area of injury.[13] That does seem logical - fewer stem cells, not as good or rapid healing, hence chronic injury. When more stem cells are added to the mix, healing occurs more rapidly.[14]
It has even been suggested that, with the proper porous joint prostheses, the addition of stem cells to the surgical procedure might improve the incorporation of the prosthesis into the patient's own body.[15] Are we seeing the dawn of the era of the "bionic man"?
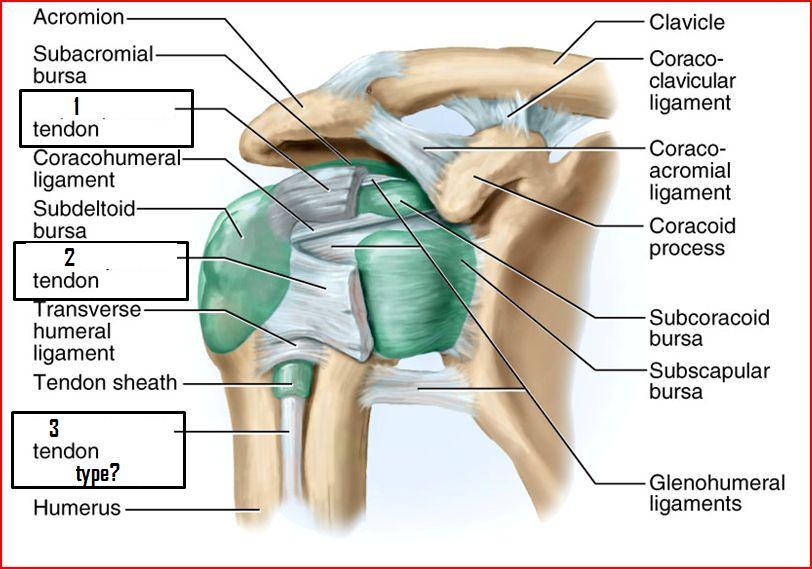 Shoulder rotator cuff injuries, for instance, are notoriously resistant to surgical repair. The addition
of biologic therapies to the surgical procedure has been recently suggested
as a way of improving the outcome.[16]
Shoulder rotator cuff injuries, for instance, are notoriously resistant to surgical repair. The addition
of biologic therapies to the surgical procedure has been recently suggested
as a way of improving the outcome.[16]
Joint injuries do not occur exclusively through accidental injury. The extent of the injury may depend on a multitude of extrinsic factors. Many of these factors are modifiable through nutrition, supplementation and lifestyle modifications. Cell apoptosis, Insulin-like Growth Factor 1 (IGF-1), nitric oxide levels, fatty infiltration, inflammatory markers - all of these are potentially modifiable factors which have an impact on overall health as well as repair from injury.
Knee joints are severely affected by osteoarthritis in many people starting at a fairly early age (at least it feels early, if you are a baby-boomer). The available treatments are symptomatic, and involve anti-inflammatory drugs and surgery to remove the damaged cartilage and inflamed bone that leads to progressively more severe cartilage loss and bone inflammation. The inevitable outcome, until now, has been either immobility or joint replacement. Not surprisingly, the health of the underlying bone has influence over the health of the joint cartilage. Resident cartilage stem cells appear to change with the change in their environment, and are unable to repair the damaged cartilage, instead producing enzymes that clean up damaged cartilage but that do not lay down new healthy cartilage. It is an inflammatory process gone awry. Mesenchymal stem cells taken from outside the joint can suppress inflammation and lead to healing of the cartilage. This has been demonstrated both in animal studies and also in a small human study.[17] The patients' most painful knee joint was injected with a dose of 8-9 x 106 cells suspended in about 1 tsp of saline solution. The less painful joint was not touched. At follow up 5 years later there had been some worsening as compared with their results at 6 months, but no knee was worse than at baseline, and all had improved at least somewhat in function. The untreated knees were all worse than at baseline. No other interventions were attempted with these patients - only injection of adipose-derived (and cultured) stem cells into the joint.
Stem cell therapy has been used for conditions previously thought to be irreversible.[18]
Adult respiratory distress syndrome, for instance - the kind where the patient is on a ventilator and their organs are starting to fail - has a dismal prognosis. Many of these patients die sooner rather than later of their lung disease - 60% in one study[19] - generally without ever getting off the ventilator.
Congestive heart failure is another condition which has no cure in allopathic medicine. We can treat the symptoms, but conventional wisdom states that heart muscles do not regenerate. Nevertheless, research shows that the heart can indeed repair itself.[20] Mesenchymal stem cells home to the area of injury, releasing healing and growth-stimulating factors for repair of the damaged heart.[21] Admittedly this latter study was in the mouse heart, not the human. This procedure is being performed on hopeful patients in stem cell clinics throughout the world at the present date. Clinical trials are currently being conducted in Europe because, as the Mayo Clinic reports, "Why European trials first? It’s faster to get many clinical studies approved in Europe compared with the United States."[22]
Wounds and radiation burns are an area where stem cells could be used to excellent effect. Radiation burns, for instance, are notoriously difficult to heal. Stem cell therapy seems like a good therapy to add to the usual treatment of such injuries.[23] , [24], [25]
Stem cells can also enhance the healing of other notoriously difficult skin wounds secondary to poor blood supply, as occurs in diabetics and those with arteriosclerosis. [26] These so-called ischemic wounds often lead to amputation, when they become gangrenous. They are treated "conservatively" with dressing changes, surgical removal of dead tissue (debridement), and eventual skin grafts. When all else fails, and the extremity is too far gone to salvage, amputation is the final solution. In 2009 it was estimated that 6.5 million patients in the United States are affected annually, with a cost of in excess of 25 billion dollars.[27] Chronic wounds - including the bed sores that afflict so many immobile patients in nursing homes - are seldom seen in healthy patients. The very reason for which wounds do not heal has to do with tissue nutrition, mitochondrial dysfunction, circulating levels of glucose and insulin, and other chronic tissue deficiencies which are relatively easy to regulate, given the correct functional medicine therapies. These illnesses are generally treated with pharmaceutical drugs - blood pressure medications, lipid-lowering drugs, blood sugar lowering drugs and the like - which treat the immediate abnormality but which are not typically known to correct the underlying metabolic deficiencies or toxicities.
Once the underlying nutritional deficiencies are corrected, and the underlying toxicities are decreased, then stem cell therapy would seem a logical choice, when conservative therapies fail, before it becomes necessary to contemplate skin grafts or tissue flaps or even amputation.[28]
What do stem cells require for development and growth?
In order to use stem cells for treatment, three requirements must be met:
Senior adult with scar on knee after surgery with knee replacement
Consider that painful knee joint which has gradually taken over your life.
You probably started by taking anti-inflammatory medicine and wearing
a knee brace. Then maybe you were prescribed physical therapy. Then maybe
steroid injections. If you were lucky, somewhere along the way acupuncture
was recommended. You might even have figured out on your own that every time you
 ate hot peppers or tomatoes, you knee started to hurt.
ate hot peppers or tomatoes, you knee started to hurt.
After a while, the knee became so painful and swollen that you were recommended to have surgery. It started out with arthroscopic surgery - looking inside the joint and removing any stray floating bits of cartilage which may have been irritating. When that didn't really help, you were told that you had "bone on bone", that you would never recover, and that your best option was a knee replacement.
You can easily see that between the initial painful experience and the need for surgery to replace the joint, there is a lot of room for considering other therapies. This is where stem cell therapy and natural medicine come in to play.
Activated platelets release growth factors like vascular endothelial growth factor (VEGF) and stromal cell derived factor (SDF-1) which then call stem cells or progenitor cells to the area of injury. There is an optimal concentration of platelets for stimulation of healing.[29] Too few and stimulation of growth is diminished. Too many and stimulation of growth is actively inihibited.

These growth factors bind to the cell membrane and in turn activate genes that encourage cell division (mitosis).[30], [31]
Platelets before activation (right)
The choice of local anesthesia may influence the effectiveness of platelet rich plasma.[32]
Platelet Rich Plasma injections are used for the following conditions:
There are disadvantages in the use of PRP. Since the platelets cause the
systemic release of growth factors, including those banned for therapeutic
use in professional athletes, enumerated on the World Anti-Doping Agency
(WADA) prohibited list[33], the use of PRP, although a "natural" form of therapy, is not
one which is available to the professional athlete if they wish to return
to competition.
How healthy are our stem cells? Much of their health depends on how well we provide for them - our diet, our lifestyle, our toxicities.[34], [35], [36] They are depleted and eventually incapacitated by products of oxidative stress (called "advanced glycation endproducts" or AGEs) commonly found in metabolic diseases like diabetes.[37]
Stem cells are regulated by their mitochondria.[38] Good mitochondrial function, good stem cells. The only problem is that stem cells, in order to stay stem cells, have to stay undifferentiated, meaning that their mitochondria have to stay quiescent - not dividing, meaning that there must be minimal oxidative stress, since oxidative stress is what pushes the stem cells into differentiation.
We see a 25-30% increase in numbers after intense physical stress (e.g. 1 hour of running) or after ingestion of certain neutraceuticals like fucoidans from seaweed - with fairly rapid return to baseline within a few hours to days.
Acupuncture has been shown to promote differentiation of bone marrow mesenchymal stem cells and functional recovery of rats with spinal cord injury.[39] Acupuncture has been shown to promote functional recovery from post-stroke injury.[40] Stem cells have been injected into acupuncture points (on dogs), with good results.[41]
A new circulatory system has been described in the literature, relating to acupuncture channels and points.[42] It is called the Bonghan system, named after the man who originally discovered it. In recent years, the network has come to be called the Primo Vascular System. Indeed, this system may well develop even before organs begin to develop - suggesting that it may be the template upon which our bodies are constructed as the embryo develops.[43]
This system has several sub-networks, including superficial (in the skin), intravascular (inside the veins, arteries and lymphatics, floating, not adherent to the vessel walls), extravascular (adhering to the outside of the blood and lymph vessels), organ surface[44] (on the membranes surrounding our internal organs), intraorgan (inside the organs themselves) and neural[45] (in the brain and spinal cord, and along the exterior of peripheral nerves). These channels and ducts were finally distinguished from lymphatic vessels by staining them with a specific dye called trypan blue - which, ironically, is also used in cell counters to distinguish active from non-active stem cells.
Here is a link to see a primo duct in a living organism: https://www.youtube.com/watch?v=JsFoTEpFpRQ
Bonghan channels appear to contain a significant number of white blood cells. Mesenchymal stem cell markers are strongly expressed in these Bonghan channels, suggesting some supportive evidence for the beneficial effects of acupuncture for immune-related diseases. Perhaps acupuncture might well be able to improve the benefits of stem cell therapy, particularly as it relates to adipose derived mesenchymal stem cells. The channels contain a high concentration of hyaluronic acid, responsible for cell growth and differentiation.[46]
Magnetic energy delivered through a pulsed magnetic frequency generator may cause stem cells to differentiate into myocardial cells.[47] Treatment with magnetic energy might eliminate the need for manipulation of cells in culture, with all the attendant risks and potential for abnormal differentiation. Stimulation with direct current at specific frequencies can cause stem cells to differentiate into neural cells, and migrate to the site of injury.[48]
Other modalities - neutraceuticals, botanicals, vitamins, antioxidants and other nutrients:
Zen stones balance against a blue horizon
Balanced oxidative stress is healthy - as it is for growing strong bones.
People who are bedridden do
 not grow straight bones. People who are under constant low level stress
have constant release of stem cells. Initially this may be seen as a good
thing - many people function better with some stress. But eventually the
system wears out. The stress is excessive, reactive oxygen species accumulate,
and they develop chronic illness, chronic fatigue, fibromyalgia, or cancer
as an ongoing expression of excessive cellular growth and reactivity.
not grow straight bones. People who are under constant low level stress
have constant release of stem cells. Initially this may be seen as a good
thing - many people function better with some stress. But eventually the
system wears out. The stress is excessive, reactive oxygen species accumulate,
and they develop chronic illness, chronic fatigue, fibromyalgia, or cancer
as an ongoing expression of excessive cellular growth and reactivity.
It is suggested that reactive oxygen species may serve as a rheostat for stem cells[70], with low levels keeping the stem cells quiescent, higher levels resulting in secretion of growth factors activate the stem cells, and excessively high levels causing the cells to commit apopotosis (cellular suicide).[71] Heavy exercise for an hour will stimulate the production of reactive oxygen species, and hence stimulate the release of stem cells into the circulation. Heavy exercise for many hours may cause the runner to "hit a wall" and be functionally incapacitated for months or years thereafter.
We live in such a finely balanced and tuned system. Stem cells require low oxygen conditions to stay quiescent, or not dividing. They decrease their oxygen consumption by stopping oxidative phosphorylation that produces a lot of energy, and switching to glycolytic metabolism (fermentation) which produces much less energy. This process is regulated by a transcription substance called HIF (hypoxia inducible factor). In order to stay quiescent, stem cells drop their oxygen requirements. Cancer cells start out by switching their metabolism under conditions of prolonged oxidative stress, so that they don't have to die. Are they trying to become stem cells again? Only their genes forget how to turn off?
Sci Rep. 2013; 3: 1978. Published online 2013 Jun 13. doi: 10.1038/srep01978. Copyright © 2013, Macmillan Publishers Limited. Image downloaded by permission, through Creative Commons License.
Rejuvenate the stem calls
It may be possible to re-introduce SIRT-3 and SIRT-7 to reverse mitochondrial
protein folding stress, the origin of cellular damage that causes stem
cells (at least hematopoietic stem cells) to age.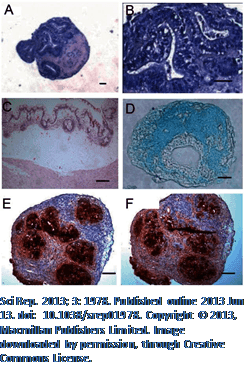 [72]
,
[73]
[72]
,
[73]
However, this is not without risk of mutation of cell lines, particularly when we are talking about induced pluripotent stem cells.[74], [75]
In my opinion, we might just be better off getting our bodies healthy, and then using our own stem cells which have not had multiple cycles of differentiation - because they are stem cells, and not stem cells forced in to an unnatural state of being. We can help our own bodies to get healthy, by putting healthy fuel into them (food) and by replenishing those nutrients which we know to be lacking (through functional nutrient testing), and then harvesting our own stem cells for our own use.
Evidence suggests that the health of mesenchymal stem cells can be improved by improving their redox status. For instance, a dysregulation of LMNA, a protein linked to poor cartilage production and resulting osteoarthritis, demonstrates excessive oxidative stress in the mitochondria. Defects in cartilage production can be partially reversed by treating the oxidative stress.[76]
Since stem cells apparently are able to "feel" the topography of their environment[77], [78], it only makes sense that if they encounter cell walls that are particularly stiff, they might "conclude" that the material is bone, and therefore begin to differentiate into osteoblasts - cells that lay down bone. It is even conceivable that part of the mechanism for laying down atherosclerotic plaque in blood vessels may be stimulated by scarring caused by hard-walled red blood cells damaging hard-walled blood vessels.
If they find tissues which are under high oxidative stress, they may conclude that it is time to multiply and divide, and divide, and divide... and perhaps lost the ability to stop dividing.[79]
Recreate new stem cells
Embryonic stem cell research is big in the scientific community. The advantages are that embryonic stem cells are either pluripotent or possibly even totipotent, so that they can be persuaded to differentiate into any cell of the body, depending upon the context in which they find themselves - whether in a wounded leg, or simply in a petri dish containing "wounded leg" compounds.
When should we NOT use stem cell therapy?
Do stem cells cause cancer? That is the big question. Is it not possible that stem cells, being capable of differentiating into anything, might simply choose, for reasons yet unknown to us, to differentiate into a cancer?
This is not an irrational argument. We see cancers in small children, who are much closer to the pluripotent state than their more differentiated adult parents. These cancers typically include those tissues which are closest to embryonic differentiation - retinoblastoma (tumor of the eye), neuroblastoma (tumor of the adrenal glands), leukemia (tumor of the blood), Wilms tumor (tumor of the kidney), and lymphoma (tumor of the immune system). We rarely see tumors of the lung, the colon, the pancreas - all organs which develop cancers in response to what we throw at them throughout our lives.
One review article outlined the evidence of parallels between abnormal metabolic factors involved with the regulation of growth in cancer cells and the same abnormal metabolic factors which occur during the restoration of human stem cells back to their pluripotent state in the laboratory, the so-called induced pluripotent stem cells, or iPSC. [80] When we manipulate living cells in the laboratory, especially when we insert virus-carried genes that regulate growth and division, we are treating these living cells just the way we treat living plant cells when we develop GMO soy, corn, zucchini, cotton - using viruses to manipulate their genetic structure. Such treatment may have far-reaching implications that we may not like. Some researchers caution that we should be working on "the development of derivation methods that produce hPSC lines that more faithfully recapitulate the normal, non-cancerous pluripotent state is needed."
To substantiate their concern, the authors reference a study which induced the formation of pluripotent stem cells, differentiated them into cartilage forming cells, and then grew them in culture. Four of the 21 lines developed abnormal looking cartilage cells that expressed tumor markers like CEA and CA 19-9.[81] That is an almost 20% chance of developing cells lines with high potential to become cancer cells.
Another article discussed the stability of induced pluripotent stem cells, and their propensity to develop teratomas (a form a cancer). Those researchers discovered that cells modified by incubating them with a virus that modified their DNA (called a virally-derived vector) were more likely to be unstable, and revert to a more primitive form even after they had completely differentiated in culture (into bone cells, or cartilage cells, or whatever the researchers were looking to develop). This evidence of instability, the authors of the paper warned, should be taken into account when using induced pluripotent cells for treatment of humans. The authors found that all the cells looked very similar initially, but that as they differentiated in cell culture, those cells that had been modified by the virus vector seemed to retain expression of genes associated with cancer. Their conclusion: "virally derived hiPSC (human induced pluripotent stem cells) are unlikely to generate clinical grade material suitable for transplantation therapeutics."[82]
By contrast, adipose derived minimally manipulated stem cells appear to be effective in treatment of multiple conditions, and particularly safe to use in the individual from whom they were taken.[83] Even in tissue culture, given the correct choice of medium, self-derived stem cells appear to be the safest to use therapeutically. If one wishes a child to develop nurturing characteristics as it grows, it really helps if the child itself is nurtured during its early years. It only makes sense that if one wishes cells to develop human associated proteins (rather than, for instance, cow-associated genes), one would give them human-associated nutrients (i.e. human serum, rather than beef serum) as they are being cultured.[84]
What are the current regulations about stem cell therapy?
Stem cells have been isolated from adipose tissue in the United States since surgeons discovered that they were abundant in the fatty tissues suctioned out of the body during a liposuction procedure.
In Europe and Asia, regulatory approval has been given for mechanical devices used to process adipose tissue using minimal manipulation, for the purpose of isolation of stem cells and the stromal vascular fraction. Therapeutic use is therefore facilitated, and this form of therapy can be easily used in the operating room and in physician offices, depending on the application.
Method of preparation of stem cell tissue has an effect on the eventual yield of cells, and the healing potential of those cells. Adipose tissue obtained through liposuction appears to provide a much higher yield of viable stem cells than tissue obtained through ultrasound methods.[85] It appears that minimally manipulated tissue is the most healthy, under ordinary circumstances, for use in the clinic.
The definition of "minimally manipulated" is becoming tighter and tighter, as the FDA debates whether to classify tissue isolated from our own bodies as a "drug" over which they have jurisdiction. In principle it would be like debating whether a skin flap surgically lifted from our own bodies might be considered to be a drug, or whether the skin flap indeed belongs to the individual from whom it was isolated. Millions of dollars in potential pharmaceutical revenues hang in the balance, pending the answer to this question.
An article[86] published under the Mayo Clinic logo from the Center for Bioethics in Minnesota reports that "stem cell tourism" is no longer the exclusive prerogative of China or Mexico, but that the same "treatments" are now available through businesses in the United States. The clear implication of this article is that these "businesses" - which are generally physician offices - are providing an unsafe treatment for the purpose of lining their own pockets, disregarding the safety or the health of the patients whom they serve.
The major fallacy in this article is one well known to those who have studied techniques of debate. It is called the "argument from (personal) incredulity", also known as the "appeal to common sense". No sensible person would believe these statements, therefore they must be false.
The article implies by judicious use of quotation marks, without any evidence of proof, that those physicians who perform stem cell therapeutic interventions in their clinics are defying the FDA by claiming that they are practicing medicine, not administering drugs, and that the FDA does not regulate the practice of medicine, only the development and safety of drugs.
The assertion that patient choice is to be decried, and the assertion that FDA bureaucratic approaches are impeding the development of stem cell therapeutics is untrue play big in the implications of the article.
The passage from the FDA regulations regarding stem cell interventions is the following:
So... is the issue compliance with FDA guidelines? Or is the issue a "turf war" between institutions wishing to be on the forefront with research and practitioners wishing to provide cutting edge therapies to their patients? Or is the issue financial, an unstated struggle between stem cells as body parts vs stem cells as drugs?
It is possible that all these factors obtain. The FDA certainly has a fluid sense of what it writes in its guidelines. At the same time, to interpret guidelines as strict rules goes counter to what the FDA itself states in the guidelines. And the third question is really one of philosophy. Are body parts in fact things that can be patented and sold on the open market? Or are body parts things that belong to the individual, who can choose to do with them what they see fit?
The guidelines are quite clear. Body parts that are minimally manipulated do not fall under the regulation of the FDA. The question then comes down to the definition of minimal manipulation, and how much manipulation causes a body part to qualify as a drug? Stay tuned as the battle rages...
What training is involved for physicians who choose to add stem cell therapy to their tool kit?
Transition to clinical practice is another issue.[87] How much training does it take to learn how to do a bone marrow aspiration, or to learn how to do liposuction of half a cup of fat from under the skin of the abdomen?
As residents, physicians are taught on the principle of "See one, do one, teach one." Physicians are expected to learn quickly, and to be dexterous with their hands, and to be honest in the evaluation of their own skills.
In plastic surgery, the difficult part is making sure that the cosmetic result is acceptable - symmetrical and adding to the beauty and/or the functionality of body part in question.
In stem cell therapy, the difficult part is learning how to inject a joint, or how to administer the end product safely. All the procedures are learned in the course of a residency, or in cadaver labs, or by observation of those who have done the procedures before, through intensive study of the body part involved, and eventually through the generosity of friends and relations who volunteer to have the procedure performed on them.
In the end, physicians must learn to do procedures by simply doing them. And those who choose to have the procedure must decide whether not having the procedure will lead to a worse result than having it.
Bioethics seems to have more to do with finances than they do with potentially therapeutic interventions for patients, utilizing their own tissues for regeneration and promotion of health. There is evidence that cultured and manipulated differentiated cells, when forced (or induced) to revert to an earlier pluripotent state, may also express genes associated with the lack of regulatory control demonstrated by cancer cells. There is evidence that use of an individual's own stem cells, without trying to induce them to do anything except simply stay alive long enough to get back into the person's body, is quite safe, and that the stem cells do not have any excess risk of turning into unregulated cancer cells.
Until the FDA shuts down those clinics providing autologous stem cell therapies, these therapies will continue to be available to patients who may benefit from them. More clinics are opening every day, promoting the use of these therapies. Some who do the procedure are more skilled than others, some are more ethical than others, some may be money-hungry, some are genuinely interested in providing the most advanced and safest tools for healing to their patients.
As with any surgical procedure, whether the procedure is relatively new or has been performed for years, the final choice is up to the patient.
So... stem cell therapy - Fountain of Youth? The Scam of the Century? Do your research, and make your own decision.
[1] Van Servellen A, Oba I. http://www.researchtrends.com/issue-36-march-2014/stem-cell-research/
[2] Ramalho-Santos M, Willenbring H. On the Origin of the Term "Stem Cell". Cell Stem Cell 1;1:35-38 (7 June 2007).
[3] Mouiseddine M1, François S et al. Human mesenchymal stem cells home specifically to radiation-injured tissues in a non-obese diabetes/severe combined immunodeficiency mouse model. Br J Radiol. 2007 Sep;80 Spec No 1:S49-55.
[4] Martin GR. Isolation of a pluripotent cell line from early mouse embryos cultured in medium conditioned by teratocarcinoma stem cells. Proc Natl Acad Sci U S A. 1981 Dec; 78(12): 7634-7638.
[5] Ammann AJ et al. Successful bone marrow transplantation in a patient with humoral and cellular immunity deficiency. Clin Exp Immunol. 1970 Sep; 7(3): 343-353.
[6] Thomas ED, Lochte HL et. al. Supralethal Whole Body Irradiation And Isologous Marrow Transplantation in Man. J Clin Invest. 1959 Oct; 38(10 Pt 1-2): 1709-1716.
[7] Published on Jan 15, 2014. MIT 7.013 Introductory Biology, Spring 2011. View the complete course: http://ocw.mit.edu/7-013S11. Instructor: Hazel Sive
[8] Hao Q, Zhu Y-G et al. Study of Bone Marrow and Embryonic Stem Cell-Derived Human Mesenchymal Stem Cells for Treatment of Escherichia coli Endotoxin-Induced Acute Lung Injury in Mice. Stem Cells Trans Med May 2015 sctm.2015-0006. doi: 10.5966/sctm.2015-0006.
[9] Dominici M, Le Blanc K et al. Minimal criteria for defining multipotent mesenchymal stromal cells. The International Society for Cellular Therapy position statement. Cytotherapy. 2006;8(4):315-7.
[10] Mills SJ, Cowin AJ, Kaur P. Pericytes, Mesenchymal Stem Cells and the Wound Healing Process. Cells 2013, 2(3), 621-634; doi:10.3390/cells2030621.
[11] Factor D1, Dale B. Current concepts of rotator cuff tendinopathy. Int J Sports Phys Ther. 2014 Apr;9(2):274-88.
[12] Stem Cell Treatment: Out from the Shadows, Onto the Cutting Edge. Downloaded 5/31/2015.
[13] Hernigou P1, Merouse G et al. Reduced levels of mesenchymal stem cells at the tendon-bone interface tuberosity in patients with symptomatic rotator cuff tear. Int Orthop. 2015 Mar 12.
[14] Hernigou P1, Flouzat Lachaniette CH et al. Biologic augmentation of rotator cuff repair with mesenchymal stem cells during arthroscopy improves healing and prevents further tears: a case-controlled study. Int Orthop. 2014 Sep;38(9):1811-8. doi: 10.1007/s00264-014-2391-1.
[15] Lewallen EA1, Riester SM et al. Biological strategies for improved osseointegration and osteoinduction of porous metal orthopedic implants. Tissue Eng Part B Rev. 2015 Apr;21(2):218-30. doi: 10.1089/ten.TEB.2014.0333.
[16] Randelli P1, Randelli F et al. Regenerative medicine in rotator cuff injuries. Biomed Res Int. 2014;2014:129515. doi: 10.1155/2014/129515.
[17] Davatchi F, Sadeghi Abdollahi B et al. Mesenchymal stem cell therapy for knee osteoarthritis: 5 years follow-up of three patients. Int J Rheum Dis 2015.
[18] Osti L, Buda M, Del Buono A. Fatty infiltration of the shoulder: diagnosis and reversibility. Muscles Ligaments Tendons J. 2014 Feb 24;3(4):351-4.
[19] Sharif N, Ifran M et al. Factors Associated within 28 Days In-Hospital Mortality of Patients with Acute Respiratory Distress Syndrome. BioMed Research International 2013, Article ID 564547. http://dx.doi.org/10.1155/2013/564547
[20] Windmolders S1, De Boeck A et al. Mesenchymal stem cell secreted platelet derived growth factor exerts a pro-migratory effect on resident Cardiac Atrial appendage Stem Cells. J Mol Cell Cardiol. 2014 Jan;66:177-88. doi: 10.1016/j.yjmcc.2013.11.016.
[21] Yan B1, Singla RD et al. Regulation of PTEN/Akt pathway enhances cardiomyogenesis and attenuates adverse left ventricular remodeling following thymosin β4 Overexpressing embryonic stem celltransplantation in the infarcted heart. PLoS One. 2013 Sep 24;8(9):e75580. doi: 10.1371/journal.pone.0075580.
[22] http://www.mayo.edu/research/discoverys-edge/regenerating-heart-tissue-stem-cell-therapy Downloaded 6/1/2015.
[23] Benderitter M, Gourmelon P et al. New emerging concepts in the medical management of local radiation injury. Health Phys. 2010 Jun;98(6):851-7. doi: 10.1097/HP.0b013e3181c9f79a.
[24] Bey E, Prat M et al. Emerging therapy for improving wound repair of severe radiation burns using local bone marrow-derived stem cell administrations. Wound Repair Regen. 2010 Jan-Feb;18(1):50-8. doi: 10.1111/j.1524-475X.2009.00562.x.
[25] Riccobono D, Agay D, et al. Application of adipocyte-derived stem cells in treatment of cutaneous radiation syndrome. Health Phys. 2012 Aug;103(2):120-6. doi: 10.1097/HP.0b013e318240595b.
[26] Park IS, Chung PS, Ahn JC. Enhancement of Ischemic Wound Healing by Spheroid Grafting of Human Adipose-Derived Stem Cells Treated with Low-Level Light Irradiation. PLoS One. 2015 Jun 11;10(6):e0122776. doi: 10.1371/journal.pone.0122776.
[27] Sen CK, Gordillo GM et al. Human Skin Wounds: A Major and Snowballing Threat to Public Health and the Economy. Wound Repair Regen. 2009 Nov-Dec; 17(6): 763-771. doi: 10.1111/j.1524-475X.2009.00543.x
[28] Duscher D, Barrera J et al. Stem Cells in Wound Healing: The Future of Regenerative Medicine? A Mini-Review. Gerontology. 2015 May 29.
[29] Giusti I, Rughetti A et al. Identification of an optimal concentration of platelet gel for promoting angiogenesis in human endothelial cells. Transfusion. 2009 Apr;49(4):771-8. doi: 10.1111/j.1537-2995.2008.02033.x.
[30] Machlus KR, Italiano JE. The incredible journey: From megakaryocyte development to platelet formation. J Cell Biol. 2013 Jun 10; 201(6): 785-796. doi: 10.1083/jcb.201304054
[31] Billaud M, Lohman AW et al. Regulation of Cellular Communication by Signaling Microdomains in the Blood Vessel Wall. Pharmacol Rev. 2014 Apr; 66(2): 513-569. doi: 10.1124/pr.112.007351It has become increasingly clear that the accumulation of proteins in specific regions of the plasma membrane can facilitate cellular communication. These regions, termed signaling microdomains, are found throughout the blood vessel wall where cellular communication, both within and between cell types, must be tightly regulated to maintain proper vascular function. We will define a cellular signaling microdomain and apply this definition to the plethora of means by which cellular communication has been hypothesized to occur in the blood vessel wall. To that end, we make a case for three broad areas of cellular communication where signaling microdomains could play an important role: 1) paracrine release of free radicals and gaseous molecules such as nitric oxide and reactive oxygen species; 2) role of ion channels including gap junctions and potassium channels, especially those associated with the endothelium-derived hyperpolarization mediated signaling, and lastly, 3) mechanism of exocytosis that has considerable oversight by signaling microdomains, especially those associated with the release of von Willebrand factor. When summed, we believe that it is clear that the organization and regulation of signaling microdomains is an essential component to vessel wall function.
[32] Bausset O, Magalon J et al. Impact of local anaesthetics and needle calibres used for painless PRP injections on platelet functionality. MLTJ. 2014 Jan-Mar; 4(1): 18-23.
[33] http://www.usada.org/substances/prohibited-list/
[34] Scheubel RJ et al. Depression of progenitor cell function by advanced glycation endproducts (AGEs): potential relevance for impaired angiogenesis in advanced age and diabetes. Exp Gerontol. 2006 May;41(5):540-8.
[35] Guilak F et al. Control of stem cell fate by physical interactions with the extracellular matrix. Cell Stem Cell. Jul 2, 2009; 5(1): 17-26.
[36] Mansilla E, Diaz Aquino V et al. Could Metabolic Syndrome, Lipodystrophy, and Aging Be Mesenchymal Stem Cell Exhaustion Syndromes? Stem Cells Int. 2011; 2011: 943216. doi: 10.4061/2011/943216
[37] Scheubel RJ et al. Depression of progenitor cell function by advanced glycation endproducts (AGEs): potential relevance for impaired angiogenesis in advanced age and diabetes. Exp Gerontol. 2006 May;41(5):540-8.
[38] Rehman J. Empowering self-renewal and differentiation: the role of mitochondria in stem cells. J Mol Med (Berl). 2010 Oct;88(10):981-6. doi: 10.1007/s00109-010-0678-2.
[39] Ding Y, Yan Q et al. Electro-acupuncture promotes survival, differentiation of the bone marrow mesenchymal stem cells as well as functional recovery in the spinal cord-transected rats. BMC Neurosci. 2009; 10: 35.
[40] Yu H, Chen PD et al. Electro-acupuncture at Conception and Governor vessels and transplantation of umbilical cord blood-derived mesenchymal stem cells for treating cerebral ischemia/reperfusion injury. Neural Regen Res. 2014 Jan 1; 9(1): 84-91. doi: 10.4103/1673-5374.125334.
[41] Marx C, Dorenelles Silveira M et al. Acupoint Injection of Autologous Stromal Vascular Fraction and Allogeneic Adipose-Derived Stem Cells to Treat Hip Dysplasia in Dogs. Stem Cells Int. 2014; 2014: 391274. doi: 10.1155/2014/391274.
[42] Soh KS. Bonghan Circulatory System as an Extension of Acupuncture Meridians. Journal of Acupuncture and Meridian Studies 2; 2: (June 2009) 93-106. doi:10.1016/S2005-2901(09)60041-8
[43] Lee SY, Lee BC, Soh KS, Jhon GJ. Development of the Putative Primo Vascular System Before the Formation of Vitelline Vessels in Chick Embryos. The Primo Vascular System 2012, pp 77-82.
[44] Lee HS, Lee JY et al. Evidence for the Primo Vascular System above the Epicardia of Rat Hearts. Evid Based Complement Alternat Med. 2013; 2013: 510461. doi: 10.1155/2013/510461 PMCID: PMC3762136
[45] Park ES, Kim HY, Young DH. The Primo Vascular Structures Alongside Nervous System: Its Discovery and Functional Limitation. Evid Based Complement Alternat Med. 2013; 2013: 538350. Published online 2013 March 31. doi: 10.1155/2013/538350 PMCID: PMC3626220
[46] Lee C, Seol SK et al. Alcian blue staining method to visualize bonghan threads inside large caliber lymphatic vessels and x-ray microtomography to reveal their microchannels. Lymphat Res Biol. 2006;4(4):181-90.
[47] Ventura C. CAM and Cell Fate Targeting: Molecular and Energetic Insights into Cell Growth and Differentiation. Evid Based Complement Alternat Med. 2005 Sep; 2(3): 277-283. doi: 10.1093/ecam/neh100
[48] Zhao H, Steiger A, Nohner M, Ye H. Specific Intensity Direct Current (DC) Electric Field Improves Neural Stem Cell Migration and Enhances Differentiation towards βIII-Tubulin+ Neurons. PLoS One. 2015 Jun 11;10(6):e0129625. doi: 10.1371/journal.pone.0129625.
[49] Nakada D, Oguro H. Oestrogen increases haematopoietic stem-cell self-renewal in females and during pregnancy. Nature. 2014 Jan 23;505(7484):555-8. doi: 10.1038/nature12932.
[50] Antoniou J, Wang HT et al. The Effects of Naproxen on Chondrogenesis of Human Mesenchymal Stem Cells. Tissue Eng Part A. 2015 May 18.
[51] Alaseem AM, Madiraju P et al. Naproxen induces type X collagen expression in human bone-marrow-derived mesenchymal stem cells through the upregulation of 5-lipoxygenase. Tissue Eng Part A. 2015 Jan;21(1-2):234-45. doi: 10.1089/ten.TEA.2014.0148.
[52] Almaawi A, Wang HT et al. Effect of acetaminophen and nonsteroidal anti-inflammatory drugs on gene expression of mesenchymal stem cells. Tissue Eng Part A. 2013 Apr;19(7-8):1039-46. doi: 10.1089/ten.TEA.2012.0129.
[53] Kudo C1, Kori M, et al. Diclofenac inhibits proliferation and differentiation of neural stem cells. Biochem Pharmacol. 2003 Jul 15;66(2):289-95.
[54] Solis MA, Chen YH et al. Hyaluronan Regulates Cell Behavior: A Potential Niche Matrix for Stem Cells. Biochemistry Research International 2012, Article ID 346972. http://dx.doi.org/10.1155/2012/346972
[55] Kim SK1, Joe Y et al. Resveratrol induces hepatic mitochondrial biogenesis through the sequential activation of nitric oxide and carbon monoxide production. Antioxid Redox Signal. 2014 Jun 1;20(16):2589-605. doi: 10.1089/ars.2012.5138.
[56] Chen T1, Li J et al. Activation of SIRT3 by resveratrol ameliorates cardiac fibrosis and improves cardiac function via the TGF-β/Smad3 pathway. Am J Physiol Heart Circ Physiol. 2015 Mar 1;308(5):H424-34. doi: 10.1152/ajpheart.00454.2014
[57] Mohrin M1, Shin J et al. Stem cell aging. A mitochondrial UPR-mediated metabolic checkpoint regulates hematopoietic stem cell aging. Science. 2015 Mar 20;347(6228):1374-7. doi: 10.1126/science.aaa2361.
[58] Rayamajhi N, Kim SK et cl. Quercetin Induces Mitochondrial Biogenesis through Activation of HO-1 in HepG2 Cells. Oxid Med Cell Longev. 2013; 2013: 154279. doi: 10.1155/2013/154279
[59] Davis JM1, Murphy EA et al. Quercetin increases brain and muscle mitochondrial biogenesis and exercise tolerance. Am J Physiol Regul Integr Comp Physiol. 2009 Apr;296(4):R1071-7. doi: 10.1152/ajpregu.90925.2008
[60] Gu Q1, Chen C et al. Ginkgo biloba extract promotes osteogenic differentiation of human bone marrow mesenchymal stem cells in a pathway involving Wnt/β-catenin signaling. Pharmacol Res. 2015 Apr 23;97:70-78. doi: 10.1016/j.phrs.2015.04.004.
[61] Wang JW, Chen W, Wang YL. A ginkgo biloba extract promotes proliferation of endogenous neural stem cells in vascular dementia rats. Neural Regen Res. 2013 Jun 25; 8(18): 1655-1662. doi: 10.3969/j.issn.1673-5374.2013.18.003
[62] Liu F, Rainosek SW et al. Protective effect of acetyl-L-carnitine on propofol-induced toxicity in embryonic neural stem cells. Neurotoxicology. 2014 May;42:49-57. doi: 10.1016/j.neuro.2014.03.011.
[63] Palermo V1, Falcone C, et al. Acetyl-L-carnitine protects yeast cells from apoptosis and aging and inhibits mitochondrial fission. Aging Cell. 2010 Aug;9(4):570-9. doi: 10.1111/j.1474-726.2010.00587.x.
[64] Lu Q, Zhang Y, Elisseeff JH. Carnitine and acetylcarnitine modulate mesenchymal differentiation of adult stem cells. J Tissue Eng Regen Med. 2013 Apr 29. doi: 10.1002/term.1747.
[65] Rehman J. Empowering self-renewal and differentiation: the role of mitochondria in stem cells. J Mol Med (Berl). 2010 Oct;88(10):981-6. doi: 10.1007/s00109-010-0678-2.
[66] Ito K, Suda T. Metabolic requirements for the maintenance of self-renewing stem cells. Nat Rev Mol Cell Biol. 2014 Apr; 15(4): 243-256. doi: 10.1038/nrm3772.
[67] Someya S, Yu W et al. Sirt3 mediates reduction of oxidative damage and prevention of age-related hearing loss under caloric restriction. Cell. 2010 Nov 24;143(5):802-12. doi: 10.1016/j.cell.2010.10.002.
[68] Jeong SG, Cho GW. Endogenous ROS levels are increased in replicative senescence in human bone marrow mesenchymal stromal cells. Biochem Biophys Res Commun. 2015 May 15;460(4):971-6. doi: 10.1016/j.bbrc.2015.03.136.
[69] Yang H, Mohamed ASS, Xhou SH. Oxidized low density lipoprotein, stem cells, and atherosclerosis. Lipids Health Dis. 2012; 11: 85. doi: 10.1186/1476-511X-11-85
[70] Liang R, Ghaffari S. Stem Cells, Redox Signaling, and Stem Cell Aging. Antioxid Redox Signal. 2014 Apr 20; 20(12): 1902-1916. doi: 10.1089/ars.2013.5300
[71] Yanes O, Clark J et al. Metabolic oxidation regulates embryonic stem cell differentiation. Nature Chemical Biology 6, 411-417 (2010). doi:10.1038/nchembio.364
[72] Shin JY, Mohrin M, Chen D. Reversing Stem cell aging. Downloaded from WWW.impactjournals.com/oncotarget 6/10/2015
[73] Mohrin M1, Shin J et al. Stem cell aging. A mitochondrial UPR-mediated metabolic checkpoint regulates hematopoietic stem cell aging. Science. 2015 Mar 20;347(6228):1374-7. doi: 10.1126/science.aaa2361.
[74] Tommasi S, Zheng A et al. Mammalian cells acquire epigenetic hallmarks of human cancer during immortalization. Nucleic Acids Res. 2013 Jan 7;41(1):182-95. doi: 10.1093/nar/gks1051.
[75] Maitra A, Arking D et al. Genomic alterations in cultured human embryonic stem cells. Nature Genetics 37, 1099 – 1103 (2005). doi:10.1038/ng1631
[76] Mateos J, De la Fuente A et al. Lamin A deregulation in human mesenchymal stem cells promotes an impairment in their chondrogenic potential and imbalance in their response to oxidative stress. Stem Cell Res. 2013 Nov;11(3):1137-48. doi: 10.1016/j.scr.2013.07.004.
[77] Murphy WL, McDevitt TC, Engler AJ. Materials as stem cell regulators. Nat Mater. 2014 Jun; 13(6): 547-557. doi: 10.1038/nmat3937
[78] Teslaa T, Teitell MA. Pluripotent stem cell energy metabolism: an update. EMBO J. 2015 Jan 13;34(2):138-53. doi: 10.15252/embj.201490446.
[79] Zhou DH, Shao LJ, Spitz DR. Reactive Oxygen Species in Normal and Tumor Stem Cells. Adv Cancer Res. 2014; 122: 1-67. doi: 10.1016/B978-0-12-420117-0.00001-3.
[80] Huo JS1, Baylin SB2, Zambidis ET. Cancer-like epigenetic derangements of human pluripotent stem cells and their impact on applications in regeneration and repair. Curr Opin Genet Dev. 2014 Oct;28:43-9. doi: 10.1016/j.gde.2014.09.008.
[81] Yamashita A, Liu S et al. Cartilage tissue engineering identifies abnormal human induced pluripotent stem cells. Sci Rep. 2013;3:1978. doi: 10.1038/srep01978.
[82] Polenco JC, Ho MD, et al. Identification of unsafe human induced pluripotent stem cell lines using a robust surrogate assay for pluripotency. Stem Cells. 2013 Aug;31(8):1498-510. doi: 10.1002/stem.1425.
[83] Nishiwaki S1, Nakayama T et al. Efficacy and safety of human adipose tissue-derived mesenchymal stem cells for supporting hematopoiesis. Int J Hematol. 2012 Sep;96(3):295-300. doi: 10.1007/s12185-012-1140-8.
[84] Josh F1, Kobe K et al. Accelerated and safe proliferation of human adipose-derived stem cells in medium supplemented with human serum. J Nippon Med Sch. 2012;79(6):444-52.
[85] Oedayrajsingh-Varma MJ1, van Ham SM et al. Adipose tissue-derived mesenchymal stem cell yield and growth characteristics are affected by the tissue-harvesting procedure. Cytotherapy. 2006;8(2):166-77.
[86] Turner LG. Federal Regulatory Oversight of US Clinics Marketing Adipose-Derived Autologous Stem Cell Interventions: Insights From 3 New FDA Draft Guidance Documents. Mayo Clinic Procedings. 90;5:567-571 (May 2015)
[87] Gimble JM, Bunnell BA, Guilak F. Human adipose-derived cells: an update on the transition to clinical translation. Regen Med. 2012 Mar; 7(2): 225-235. doi: 10.2217/rme.11.119.
